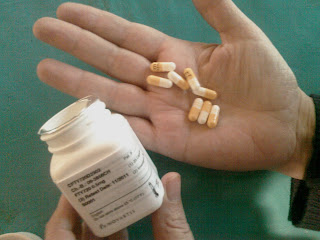
Holy CRAP!
http://www.bloomberg.com/news/2010-09-30/novartis-gilenya-ms-pill-to-cost-48-000-a-year-update1-.html
What I want to know, oh You Who Run Novartis Yet Do Not Yourself Suffer From MS, is how you people can sleep at night.
Give us a pill which will change our life -- mind you NOT CURE MS so we can be done with it, but merely give us hope and ease our suffering in such a way as to make us Fingolimod junkies for life -- and then try to make us pay $4000 a MONTH for it?? I KNEW when I saw those souped up pill container prototypes in that survey I was asked to take that you would be pulling this. Didn't I say just that?
This really ticks me off. It's the height of greed. I realize the trials were huge and cost tons of money, but production is going to be cheap. There are no syringes to supply, no medication to keep refrigerated. You know as well as I do that you will recover your testing costs within probably the first year on the market. This is just OBSCENE! I can't even begin to put into words how disappointed I am.
Do you think people with MS are just all rich or something? How dare you take advantage of our condition that way. To bring a pill to market -- sure, so you can profit, I get the goal -- but to make it so astronomically priced as to only be a cruel tease for those of us who can't afford it.
It was mentioned in the article that there are assistance programs for those who can't afford it. Well here's a novel idea you can do a clinical trial about... see how much further your drug goes to help people when you price it affordably to start with.

My story of being a hypochondriac, an MS patient, and a guinea pig. NOTE: After 16 years on Fingolimod, I'm starting my self-designed study to taper off the drug while avoiding the now well-documented "rebound relapse" phenomenon. I'll be writing about my journey on SubStack and you can find it here: https://farewellfingolimod.substack.com/
Thursday, September 30, 2010
Gilenya to cost $48000 per year!!
Saturday, September 25, 2010
It's human nature I guess
Ever wonder why the nightly news is always negative? Why we will zip right by an amazing sunset, but give us a good car wreck and we are slowing down to take in ever gory details? I guess it's only human nature to be fascinated with the ugly side of life.
I went to a message board for people with MS. Anyone who's had MS for any length of time has been there, I'm sure, as it's probably the biggest one for our condition on the web. I used to hang out there quite a bit back before the trial started, and even well into my first year of the study.
Then one day I realized there is life outside of MS and I was feeling so much better on the Fingolimod (ok, ok Gilenya) that I wasn't dwelling on my condition as much as getting back to the business of living. Once I broke away from that message board my head became more clear and my depression started to lift. I attributed it to the medication and the relief of finally having the relapses subside.
Well, yesterday I went back to visit and see if anyone was talking about the pill. I mean, I'm so giddy with excitement that I just had to see what folks were saying. I found a thread where the mention was made of the drug getting approval, and while most folks seemed hopeful, several others were quick to point out the serious side effects.
I was overcome with an urge to let everyone know just how much good it has done for me, so I posted.
Imagine my surprise and hurt when someone responded that they were tired of hearing good news and wished someone who'd had side effects would post.
Really?? Good news about the long anticipated PILL FOR MS was something they didn't want to hear?? I know I'm Novartis' #1 fan and I feel I owe them my very health as I know it today, but they ain't payin' me, people. This is real and it's my genuine experience. I haven't been blogging for the past 3 years just to string you along and lure you in so Novartis can finish you off with their pill. True, your mileage may vary, but come on! I"m not the only one out of the 1250 or so trial participants (in what has been touted as the largest clinical trial for MS EVER) to have a positive outcome.
What? My experience ALONE got the FDA to say "ya know, that patient #008 at that Florida location seems to have done really well. Ah, what the hey, let's give it a rubber stamp and call it a day."
I'm not buying it.
Especially after the Tysabri fiasco, don't you think they are crossing t's and dotting i's these days when it comes to immunomodulating drugs??
So I left a comment saying I was sorry to have been the bearer of good news and that I didn't realize they weren't looking for that.
I won't be going back. I know that a lot of great folks are there and that there is a lot of suffering going on and people looking for camaraderie, but there is also a lot of negativity. I don't need that in my life.
There are too many beautiful sunrises I want to pull over to watch. I'm not stopping to see the train wreck, and if you wanted me to tell you about it I guess you'll just have to wait for the evening news instead.
I went to a message board for people with MS. Anyone who's had MS for any length of time has been there, I'm sure, as it's probably the biggest one for our condition on the web. I used to hang out there quite a bit back before the trial started, and even well into my first year of the study.
Then one day I realized there is life outside of MS and I was feeling so much better on the Fingolimod (ok, ok Gilenya) that I wasn't dwelling on my condition as much as getting back to the business of living. Once I broke away from that message board my head became more clear and my depression started to lift. I attributed it to the medication and the relief of finally having the relapses subside.
Well, yesterday I went back to visit and see if anyone was talking about the pill. I mean, I'm so giddy with excitement that I just had to see what folks were saying. I found a thread where the mention was made of the drug getting approval, and while most folks seemed hopeful, several others were quick to point out the serious side effects.
I was overcome with an urge to let everyone know just how much good it has done for me, so I posted.
Imagine my surprise and hurt when someone responded that they were tired of hearing good news and wished someone who'd had side effects would post.
Really?? Good news about the long anticipated PILL FOR MS was something they didn't want to hear?? I know I'm Novartis' #1 fan and I feel I owe them my very health as I know it today, but they ain't payin' me, people. This is real and it's my genuine experience. I haven't been blogging for the past 3 years just to string you along and lure you in so Novartis can finish you off with their pill. True, your mileage may vary, but come on! I"m not the only one out of the 1250 or so trial participants (in what has been touted as the largest clinical trial for MS EVER) to have a positive outcome.
What? My experience ALONE got the FDA to say "ya know, that patient #008 at that Florida location seems to have done really well. Ah, what the hey, let's give it a rubber stamp and call it a day."
I'm not buying it.
Especially after the Tysabri fiasco, don't you think they are crossing t's and dotting i's these days when it comes to immunomodulating drugs??
So I left a comment saying I was sorry to have been the bearer of good news and that I didn't realize they weren't looking for that.
I won't be going back. I know that a lot of great folks are there and that there is a lot of suffering going on and people looking for camaraderie, but there is also a lot of negativity. I don't need that in my life.
There are too many beautiful sunrises I want to pull over to watch. I'm not stopping to see the train wreck, and if you wanted me to tell you about it I guess you'll just have to wait for the evening news instead.
Wednesday, September 22, 2010
Today is the day!! The FDA Approves Gilenya (Fingolimod, FTY720)
I had been watching closely all day yesterday for the mention of approval, but went to bed broken hearted.
Today was a mad house of domestic flurries and I forgot all about checking the FDA site. I even forgot to check my email all day until just now.
How fitting it was, then, that the first email I should happen upon was that of my clinical trial coordinator. She sent no message but rather a link to an article announcing the FDA approval.
I'm elated, excited, overjoyed and nearly in tears about this news. Whether tears of joy for all who have been awaiting the pill, or tears of sorrow as a chapter in my life will soon be ending and I will once again have to figure out how to pay for my meds, I'm not sure.
I feel like jumping up and down and I don't know why there's no fireworks when I look outside... surely it will be on the evening news, right?? I mean this IS BIG! The first EVER pill for MS. Wow! And I've been on it for 3 years already!
Anyhow, here is the news straight from Novartis' mouth:
http://www.novartis.com/newsroom/media-releases/en/2010/1445917.shtml
And another link on Bloomberg Business Week:
http://www.businessweek.com/news/2010-09-22/novartis-awarded-u-s-approval-for-ms-pill-gilenya.html
I guess I have to get used to saying "Gilenya" now. Took me forever to learn to spell Fingolimod. Thanks Novartis.
Really, though... THANKS NOVARTIS!! -- and CONGRATULATIONS! You gave me my life back one pill at a time. I wish you much success with lots more MSers out there.
Today was a mad house of domestic flurries and I forgot all about checking the FDA site. I even forgot to check my email all day until just now.
How fitting it was, then, that the first email I should happen upon was that of my clinical trial coordinator. She sent no message but rather a link to an article announcing the FDA approval.
I'm elated, excited, overjoyed and nearly in tears about this news. Whether tears of joy for all who have been awaiting the pill, or tears of sorrow as a chapter in my life will soon be ending and I will once again have to figure out how to pay for my meds, I'm not sure.
I feel like jumping up and down and I don't know why there's no fireworks when I look outside... surely it will be on the evening news, right?? I mean this IS BIG! The first EVER pill for MS. Wow! And I've been on it for 3 years already!
Anyhow, here is the news straight from Novartis' mouth:
http://www.novartis.com/newsroom/media-releases/en/2010/1445917.shtml
And another link on Bloomberg Business Week:
http://www.businessweek.com/news/2010-09-22/novartis-awarded-u-s-approval-for-ms-pill-gilenya.html
I guess I have to get used to saying "Gilenya" now. Took me forever to learn to spell Fingolimod. Thanks Novartis.
Really, though... THANKS NOVARTIS!! -- and CONGRATULATIONS! You gave me my life back one pill at a time. I wish you much success with lots more MSers out there.
Friday, September 10, 2010
We Were Beaten to the Punch! Russia Approves Fingolimod!
Russian Authorities Become First to Sanction Novartis’ Oral MS Therapy
GEN news highlights: Sep 10, 2010
Russian regulatory authorities have approved Novartis’ Gilenya® 0.5 mg once-daily oral treatment (FTY720; fingolimod) for relapsing remitting multiple sclerosis (MS). Russia is the first country to approve the sphingosine 1-phosphate receptor (S1PR) modulator, and Novartis plans to launch the drug in early 2011.
An FDA decision on Gilenya is expected during September, and the outlook for U.S. approval looks promising, given that the agency’s advisory committee gave the drug a unanimous thumbs up back in June. Gilenya is also under review by the European Medicines Agency and by other regulatory bodies worldwide.
Novartis says Gilenya is the first oral therapy for treating relapsing MS. The Phase III trial program confirmed the drug reduces relapses, disability progression over two years, and brain lesions. Data also showed Gilenya to be more effective than Biogen Idec’s Avonex®, the firm adds. The FDA granted Gilenya priority review status in February 2010, reducing the standard ten-month review to six months. In May, the FDA extended the priority review period by three months to September 2010.
Aside from the fact that I believe they spelled Gilenia incorrectly, this news gives me chills. This wasn't a dead end, waste of time study (not that I thought so, but I remember my hesitancy when presented with the Clinical Trial option for trying out therapy). It's really coming to pass.
They beat us by 11 days. But it's coming. I expect ticker tape parades, special treatment, and paparazzi everywhere due to the fame we are all going to have once this thing takes the MS world by storm.
Reality check: ticker tape parade is when the paper shredder goes haywire, special treatment comes when you get phone calls from bill collectors asking for you BY NAME! and the paparazzi is just someone in your family catching you having a bad hair day and deciding for some reason that it should be recorded for posterity.
I can be famous if I try hard enough... see?
GOOOOOOOOOOOOOOOOOOOOOOOO Fingo!
An FDA decision on Gilenya is expected during September, and the outlook for U.S. approval looks promising, given that the agency’s advisory committee gave the drug a unanimous thumbs up back in June. Gilenya is also under review by the European Medicines Agency and by other regulatory bodies worldwide.
Novartis says Gilenya is the first oral therapy for treating relapsing MS. The Phase III trial program confirmed the drug reduces relapses, disability progression over two years, and brain lesions. Data also showed Gilenya to be more effective than Biogen Idec’s Avonex®, the firm adds. The FDA granted Gilenya priority review status in February 2010, reducing the standard ten-month review to six months. In May, the FDA extended the priority review period by three months to September 2010.
================ ~*~ ================
Aside from the fact that I believe they spelled Gilenia incorrectly, this news gives me chills. This wasn't a dead end, waste of time study (not that I thought so, but I remember my hesitancy when presented with the Clinical Trial option for trying out therapy). It's really coming to pass.
They beat us by 11 days. But it's coming. I expect ticker tape parades, special treatment, and paparazzi everywhere due to the fame we are all going to have once this thing takes the MS world by storm.
Reality check: ticker tape parade is when the paper shredder goes haywire, special treatment comes when you get phone calls from bill collectors asking for you BY NAME! and the paparazzi is just someone in your family catching you having a bad hair day and deciding for some reason that it should be recorded for posterity.
I can be famous if I try hard enough... see?
GOOOOOOOOOOOOOOOOOOOOOOOO Fingo!
Thursday, September 9, 2010
The News I Received at my TRANSFORMS checkup 22
I didn't mean to leave you hanging -- I really did intend to finish this up, but life got in the way.
Anyway, while I was being examined by the head neurologist overseeing the study in my area, he mentioned that everyone is excited that Gilenia is on track for FDA approval to be announced on the 21st of September.
EEEEEK!! That's too soon. (Sorry to those of you so patiently waiting, I just had to have a selfish moment).
Actually this is amazing news and I am so very excited! So was the neuro. He said "After all this is over, and history is made with this new pill form of treatment, you can say you had a part in making history. What you have done will benefit MSers for years to come. You should be proud."
He also told me that Novartis has me scheduled to stay in the study for another year (translates "1 more year or drugs.") and then they will cut me loose with a 3 month supply as a parting gift.
This is totally different from what other people have claimed their trial people have told them. I thought I distinctly remembered crying tears of joy when someone said they knew for a fact we'd be getting 5 years for free.
Anyway, the drug will be a second line drug and will only be prescribed if you have failed 2 other forms of MS drug treatment. Ironically, even though I have been ON this drug for over 3 years now, I would have to prove that I can't take the others before I can be prescribed this.
So I go home floating on a cloud as the reality of how all these years of poking and prodding are resulting in a pill form of MS treatment finally coming to pass. It just makes me burst with pride at all us guinea pigs hanging in there, and I have so much hope in my heart for those of you who cling to every whisper of a hint that something new and better is just right around the corner.... It REALLY IS!
I have said it before and I'll say it again. I was in a terrible spot before this study, and while I knew I was blessed not to be as disabled as other less fortunate MSers, that thought didn't lessen my miserable attitude and feelings of hopelessness. Looking back, I think now that I may have been nearly suicidal. I was just at the end of what I thought I could deal with and knowing it couldn't get any better didn't help. (Rationally, I know now that's not true, but in my own mind, warped by depression, that was how I felt).
This study gave me my life back. I feel better and better all the time, and have not had an MS attack in over 3 years now. Back then, you couldn't have convinced me I'd be sitting here blogging these words 3 years later.
So if you are at your wit's end and praying that something will help you because you just can't take another day of your situation, please hang in there and know that something better truly is within your reach now... just a few more days...
The New Look:
After trading in my empty pill bottles for 3 new sealed bottles, I woke up the next morning after my appointment and groggily grabbed one of them to take my daily pill. I puncture the foil seal on the bottle to set the capsules free and rolled one into my hand. I stare and blink. Not comprehending.
I peer into the bottle to see it was full of about 35 more capsules that looked just like it.
I slap the first pill back inside and cap the bottle, setting it quickly on the table as if it were poison, shoving it away.
I whipped out my cell phone and call the trial nurse, who, much to her chagrin, is on my speed dial.
Trial coordinator: Hello?
Me: I just open my medicine and they've made a mistake!! I got the wrong pills!
Trial coordinator: What? I don't understand. What are you talking about?
Me: The PILLS! They aren't pink or brown, or whatever you want to call that color!
Trial coordinator: They're not??! What do they look like?
Me: (getting out bifocals and trying to really see them) Well, they are gold on one end, white on the other end, and have little gold racing stripes that go around the white end.
Trial coordinator: (silence)
Me: are you there?
Trial coordinator: oh, yes, I am here, but I am confused. Let me call Novartis and I will call you back. Wait 10 minutes and do not take the medicine.
Me: Ok (squinting) but now that I look closer I do see it says "FTY 0.5mg" on the one end of the pill.... maybe this is the new look??
Trial coordinator: That is what I suspect, but let me call to confirm first.
So I wait 10 minutes and my cell phone rings.
Trial coordinator: (excited!) Yes! That is the new look! Tell me again what they look like because now I am curious since I haven't seen them.
Me: (describes them again)
Trial coordinator: I called all the other patients and asked them to open all their bottles and look. You are the only one who got the new pills!
I wonder if it's because Novartis has been watching my blog? (I'm full of myself, aren't I?) but seriously. They have been reading my blog a lot. It's in my stats.
So, if Novartis is following along, isn't all this great publicity you're getting from my blog just wonderful? Say, worth a lifetime of pills? (sigh I had to try).
Anyhow... here they are... TA DA! Sorry the picture isn't so great but we had to use my sister's cell phone since my video camera's "camera only" mode sucks.
Now I'm sorry I didn't hold back on of the pink ones. They're gone forever all but for the picture in the background of this blog.
I guess they needed to make them gold and add racing stripes to justify the price they're going to be gouging us for.
Anyway, while I was being examined by the head neurologist overseeing the study in my area, he mentioned that everyone is excited that Gilenia is on track for FDA approval to be announced on the 21st of September.
EEEEEK!! That's too soon. (Sorry to those of you so patiently waiting, I just had to have a selfish moment).
Actually this is amazing news and I am so very excited! So was the neuro. He said "After all this is over, and history is made with this new pill form of treatment, you can say you had a part in making history. What you have done will benefit MSers for years to come. You should be proud."
He also told me that Novartis has me scheduled to stay in the study for another year (translates "1 more year or drugs.") and then they will cut me loose with a 3 month supply as a parting gift.
This is totally different from what other people have claimed their trial people have told them. I thought I distinctly remembered crying tears of joy when someone said they knew for a fact we'd be getting 5 years for free.
Anyway, the drug will be a second line drug and will only be prescribed if you have failed 2 other forms of MS drug treatment. Ironically, even though I have been ON this drug for over 3 years now, I would have to prove that I can't take the others before I can be prescribed this.
So I go home floating on a cloud as the reality of how all these years of poking and prodding are resulting in a pill form of MS treatment finally coming to pass. It just makes me burst with pride at all us guinea pigs hanging in there, and I have so much hope in my heart for those of you who cling to every whisper of a hint that something new and better is just right around the corner.... It REALLY IS!
I have said it before and I'll say it again. I was in a terrible spot before this study, and while I knew I was blessed not to be as disabled as other less fortunate MSers, that thought didn't lessen my miserable attitude and feelings of hopelessness. Looking back, I think now that I may have been nearly suicidal. I was just at the end of what I thought I could deal with and knowing it couldn't get any better didn't help. (Rationally, I know now that's not true, but in my own mind, warped by depression, that was how I felt).
This study gave me my life back. I feel better and better all the time, and have not had an MS attack in over 3 years now. Back then, you couldn't have convinced me I'd be sitting here blogging these words 3 years later.
So if you are at your wit's end and praying that something will help you because you just can't take another day of your situation, please hang in there and know that something better truly is within your reach now... just a few more days...
The New Look:
After trading in my empty pill bottles for 3 new sealed bottles, I woke up the next morning after my appointment and groggily grabbed one of them to take my daily pill. I puncture the foil seal on the bottle to set the capsules free and rolled one into my hand. I stare and blink. Not comprehending.
I peer into the bottle to see it was full of about 35 more capsules that looked just like it.
I slap the first pill back inside and cap the bottle, setting it quickly on the table as if it were poison, shoving it away.
I whipped out my cell phone and call the trial nurse, who, much to her chagrin, is on my speed dial.
Trial coordinator: Hello?
Me: I just open my medicine and they've made a mistake!! I got the wrong pills!
Trial coordinator: What? I don't understand. What are you talking about?
Me: The PILLS! They aren't pink or brown, or whatever you want to call that color!
Trial coordinator: They're not??! What do they look like?
Me: (getting out bifocals and trying to really see them) Well, they are gold on one end, white on the other end, and have little gold racing stripes that go around the white end.
Trial coordinator: (silence)
Me: are you there?
Trial coordinator: oh, yes, I am here, but I am confused. Let me call Novartis and I will call you back. Wait 10 minutes and do not take the medicine.
Me: Ok (squinting) but now that I look closer I do see it says "FTY 0.5mg" on the one end of the pill.... maybe this is the new look??
Trial coordinator: That is what I suspect, but let me call to confirm first.
So I wait 10 minutes and my cell phone rings.
Trial coordinator: (excited!) Yes! That is the new look! Tell me again what they look like because now I am curious since I haven't seen them.
Me: (describes them again)
Trial coordinator: I called all the other patients and asked them to open all their bottles and look. You are the only one who got the new pills!
I wonder if it's because Novartis has been watching my blog? (I'm full of myself, aren't I?) but seriously. They have been reading my blog a lot. It's in my stats.
So, if Novartis is following along, isn't all this great publicity you're getting from my blog just wonderful? Say, worth a lifetime of pills? (sigh I had to try).
Anyhow... here they are... TA DA! Sorry the picture isn't so great but we had to use my sister's cell phone since my video camera's "camera only" mode sucks.
Now I'm sorry I didn't hold back on of the pink ones. They're gone forever all but for the picture in the background of this blog.
I guess they needed to make them gold and add racing stripes to justify the price they're going to be gouging us for.
Wednesday, September 8, 2010
Checkup 22 TRANSFORMS FTY720, Fingolimod, Gilenia Clinical Trial
On the 20th of August I had reached the big 3 year milestone in this clinical trial and had a nagging thought that the clinical trial director had told me when my anniversary appointment would be, but I didn't worry too much because she ALWAYS calls the week before to make certain I didn't forget (which I always do).
This time was the same as usual -- on my part -- and I completely forgot. It was not the same as usual on her part as she, too, forgot. She called me yesterday as I was about to take my pill somewhere near 9am. She told me she forgot to call and all my tests were scheduled for that day. It's a 2 hour drive to get there and I hadn't showered yet, so we compromised and she rescheduled some stuff. I got her to push the dreaded MRI back 2 weeks and the skin exam will be the same day.
Whew. I CANNOT just be told "you're having an MRI now," without a sufficient period of pre-MRI worry time. I have to psych myself up for it like Rocky Balboa drinking his pitcher full of raw eggs and stuff. Only my pitcher needs to be full of Xanax.
So I get my shower and call my friend Karen to ride up with me. She drops everything and goes. I know I'll need her for the ride back if the 4:30pm eye exam goes anything like last time .
We get there and I go right in. I have my vitals taken, blood drawn, EKG and peg test and fast paced walking. Then I go across the street to the hospital for my PFT. Everywhere I go everyone is abuzz about how the med is nearing approval.
I ace all my tests and come back to the Research Department for my EDSS and the Numbers Guy. That's the one where the recorded guy says a number every 3 seconds and you have to add the first two, say the result, then forget what you said, remember the last number he said, and add it to the new number he said. Sound confusing? Try doing it.
Evil Recording guy: 5 ...... 3
Me: 8
Evil Recording guy: 7
Me: 10
and so on...
I did perfect until halfway through when I became acutely self-conscious with Karen in the room. I was wondering if she was following along and heard me hesitate at the dreaded 9's. I don't know why I get hung up adding 9 to a number but I do and always before I just got a little slower; I didn't go dead in the water with Evil Recording guy spouting off numbers while I drooled.
Yesterday I missed 3. Out of 60 numbers, that's not too bad.
Then I had my EDSS test and I got to skip walking the hall since I had walked across the street to the hospital and back and that was far enough according to the people in charge of testing me. They used a high tech way of measuring my ability. They asked Karen if I could keep up with her. Hey, I'm not complaining. Any time I get out of the "25 Lap Hall Walking Marathon" for MS I'm happy.
Then it was on to the eye exam which I had been stressing over since my incredible headache and starry-eyed vision the last time I got dilated. After a lengthy conversation with the eye doctor, she reassured me that I would be fine and we proceeded with the drops.
After the eye appointment I had to go back to Research and see the head neuro overseeing the trial at this location. He wacked me with his rubber triangle hammer in a few places, told me how I looked better and better every time I saw him and then started talking about the Exciting News!
With that teaser I'll just leave you hanging for a brief time while I tend to my duties long enough to carve out another slot of time in which I can sit and type the juicy details. Stay tuned!
This time was the same as usual -- on my part -- and I completely forgot. It was not the same as usual on her part as she, too, forgot. She called me yesterday as I was about to take my pill somewhere near 9am. She told me she forgot to call and all my tests were scheduled for that day. It's a 2 hour drive to get there and I hadn't showered yet, so we compromised and she rescheduled some stuff. I got her to push the dreaded MRI back 2 weeks and the skin exam will be the same day.
Whew. I CANNOT just be told "you're having an MRI now," without a sufficient period of pre-MRI worry time. I have to psych myself up for it like Rocky Balboa drinking his pitcher full of raw eggs and stuff. Only my pitcher needs to be full of Xanax.
So I get my shower and call my friend Karen to ride up with me. She drops everything and goes. I know I'll need her for the ride back if the 4:30pm eye exam goes anything like last time .
We get there and I go right in. I have my vitals taken, blood drawn, EKG and peg test and fast paced walking. Then I go across the street to the hospital for my PFT. Everywhere I go everyone is abuzz about how the med is nearing approval.
I ace all my tests and come back to the Research Department for my EDSS and the Numbers Guy. That's the one where the recorded guy says a number every 3 seconds and you have to add the first two, say the result, then forget what you said, remember the last number he said, and add it to the new number he said. Sound confusing? Try doing it.
Evil Recording guy: 5 ...... 3
Me: 8
Evil Recording guy: 7
Me: 10
and so on...
I did perfect until halfway through when I became acutely self-conscious with Karen in the room. I was wondering if she was following along and heard me hesitate at the dreaded 9's. I don't know why I get hung up adding 9 to a number but I do and always before I just got a little slower; I didn't go dead in the water with Evil Recording guy spouting off numbers while I drooled.
Yesterday I missed 3. Out of 60 numbers, that's not too bad.
Then I had my EDSS test and I got to skip walking the hall since I had walked across the street to the hospital and back and that was far enough according to the people in charge of testing me. They used a high tech way of measuring my ability. They asked Karen if I could keep up with her. Hey, I'm not complaining. Any time I get out of the "25 Lap Hall Walking Marathon" for MS I'm happy.
Then it was on to the eye exam which I had been stressing over since my incredible headache and starry-eyed vision the last time I got dilated. After a lengthy conversation with the eye doctor, she reassured me that I would be fine and we proceeded with the drops.
After the eye appointment I had to go back to Research and see the head neuro overseeing the trial at this location. He wacked me with his rubber triangle hammer in a few places, told me how I looked better and better every time I saw him and then started talking about the Exciting News!
With that teaser I'll just leave you hanging for a brief time while I tend to my duties long enough to carve out another slot of time in which I can sit and type the juicy details. Stay tuned!
Sunday, September 5, 2010
Results of the Bone Density Scan and Aftermath
Last time I saw the GYN he told me my blood test revealed my ovaries had given up the ghost and were just sorry sacks of freeloading flesh, never to serve another productive day in their pathetic lives. Well, I'm paraphrasing him, but that's basically the gist of it. I have entered menopause.
He gave me a script for Estradiol that is bioidentical (or molecularly identical) to human estrogen. He also had to give me a handout with all the data gleaned from the Women's Health Initiative, a poorly design study in which skewed results were obtained due to many factors, not the least of which being that the median age of their "newly menopausal" subjects happened to be 63!?!
Being the certified hypochondriac that I am, I could not just start popping a pill just because it was prescribed for me without a lot of agonizing over the whole ordeal. I don't suffer from menopausal symptoms to such a degree that my life is disrupted by them. The main issue was going to be with whether or not it was going to help me with bone density.
I have had a LOT of steroids since being diagnosed with MS and I had strong suspicions that my bones were thin. I have other risk factors including being "thin" (that's according to my doc, not my own definition because I think I could stand to lose 10lbs), not eating enough calcium rich foods due to being lactose intolerant, I used to smoke, and I am menopausal.
So I promised myself I would hold off on the estrogen decision until the results of the bone density test came back. That happened this past Tuesday when I went in for my checkup.
The nurse looks through my file when she's preparing to take my vitals and she's the one who broke the news. "You have osteopenia. It's a precursor to osteoporosis. Here's a bunch of pamphlets to read."
Gee. Pretty anticlimactic.
So I'm going to see the doc and he's a real talker. My sister had the appointment slot 1/2 hour previous to mine and they put us in adjacent (not to be confused with adjoining) rooms. The doctor talked and talked. I got so bored waiting for my turn that I decided to go all McGyver and try to hear what was going on by cupping my hands in a circle tightly against the wall and jamming my ear down into my home made water glass and see if I could hear anything. I could.
Not much, mind you. Mostly my sisters responses because a) her voice is louder and b) she was facing the wall.
Having fun with my spy game, I decided to get all scientific about it and stand up, slowly sliding my water-glass-hand-cup along the wall searching for the optimal audio reception, finding that the studs really do hinder the sound quality, and if I went above the counter level I could get even better audio.
Realizing the compromising position I was in, half squatting with my ear plaster to my hand-cup against the wall and between the exam table and the chair they give you so your clothes aren't just thrown on the floor, I decided that I better just chill out and resume a more natural stance in a section of the room the nurse would not be alarmed to find me.
It wasn't like he'd been telling her anything she's have needed to go all HIPPA on me about. They were discussing what foods constituted a balanced diet. Big whoop. But it was something to do and I tend to do stuff that hidden camera people would EAT UP if I think I'm alone.
About the time I got situated back in my chair where I had been told to sit, stay, was about the same time I heard her door open and the doc saying his goodbyes. Then I could hear him and the nurse discussing his next patient (moi) out in the hallway.
"Oh, they're sisters? Really! Cool."
He then came in, not mentioning my sister or any Family Plan Discount Coupon he might have in his wallet, but just shook my hand and got busy reviewing my file.
He talked about osteopenia and how it shouldn't be confused with osteoporosis since that's not what I have. We talked about how I need to take an aggressive proactive stance against it and start making changes that would decrease my odds of ending up with osteoporosis.
He then asked me how the Estradiol was working for me. I told him my plan to hold off starting until I got the results he'd just given me. He said that was a fair decision. (I didn't know I was getting judged on my decision making).
So now that I was presented with the results it was do or die time. We rehashed all the risk factors checking off those which apply to me..
"Used to smoke, don't have high intake of calcium, sedentary lifestyle, you're thin..."
"Woah! What was that? You think I"m thin?? Can I get that in writing on your watermarked letterhead?"
Then we go on to discuss the percentage of body fat that people have and your optimal body fat in order to hover in a safe place farthest from osteoporosis risk (on the low end) and cardiac complications (on the high end). It needs to be between 25 and 30% on the scale.
Then he tells me there are scales which can accurately calculate body fat and body water, and you can purchase them at wal-mart.com among other places. Intrigued I whipped out some scratch paper and a pen and started taking notes.
Simultaneously he whipped out his iPhone and started surfing to find the one I needed to get, were I so inclined. After writing the brand name (Taylor) and that it needed to be digital and say that it measures body fat and body water, I felt my heart skip a beat...
I had a defective iron I had purchased the day before and needed the receipt to take it back for exchange. I was hoping against hope I wasn't scribbling all over the back of it. I flipped it over and for a second I was relieved.
Then I burst out laughing and confessed to the doctor that I was taking notes about the scale on the back of a Dunkin Donuts receipt. He, too, had a good laugh.
So we decided that I would increase my calcium intake to between 1100 and 1500 mg / day (I have a 1200 mg chewable I got from Walgreens that fits the bill), and I would get that scale, (which I ordered from Wal-mart.com from this ad) , and that I would begin taking the Estradiol as opposed to my inevitable choices I would later be presented with should my bones continue thinning...
Things like Boneva, Fosomax, and other super expensive osteo drugs. I decided I'd give the Estradiol (at $17 for a 1 mo. supply or if I join Walgreens Rx plan I can get it for $4/mo for a 3mo supply) a try. May as well try the cheap route since I'm not made of money last I checked.
That brings us up to date and I am on day 3 of being an estroginated subject.
Here's what I have found.
If this helps my menopausal symptoms I had been ignoring (the sleep issues, hot flashes and hip aches) and helps increase my bone density, then I guess I am willing to risk certain minimally possible adverse events.
After all, I'm becoming a pro at this drug taking, weigh the pros and cons stuff. I've had 3 years practice with Fingolimod. :D
He gave me a script for Estradiol that is bioidentical (or molecularly identical) to human estrogen. He also had to give me a handout with all the data gleaned from the Women's Health Initiative, a poorly design study in which skewed results were obtained due to many factors, not the least of which being that the median age of their "newly menopausal" subjects happened to be 63!?!
Being the certified hypochondriac that I am, I could not just start popping a pill just because it was prescribed for me without a lot of agonizing over the whole ordeal. I don't suffer from menopausal symptoms to such a degree that my life is disrupted by them. The main issue was going to be with whether or not it was going to help me with bone density.
I have had a LOT of steroids since being diagnosed with MS and I had strong suspicions that my bones were thin. I have other risk factors including being "thin" (that's according to my doc, not my own definition because I think I could stand to lose 10lbs), not eating enough calcium rich foods due to being lactose intolerant, I used to smoke, and I am menopausal.
So I promised myself I would hold off on the estrogen decision until the results of the bone density test came back. That happened this past Tuesday when I went in for my checkup.
The nurse looks through my file when she's preparing to take my vitals and she's the one who broke the news. "You have osteopenia. It's a precursor to osteoporosis. Here's a bunch of pamphlets to read."
Gee. Pretty anticlimactic.
So I'm going to see the doc and he's a real talker. My sister had the appointment slot 1/2 hour previous to mine and they put us in adjacent (not to be confused with adjoining) rooms. The doctor talked and talked. I got so bored waiting for my turn that I decided to go all McGyver and try to hear what was going on by cupping my hands in a circle tightly against the wall and jamming my ear down into my home made water glass and see if I could hear anything. I could.
Not much, mind you. Mostly my sisters responses because a) her voice is louder and b) she was facing the wall.
Having fun with my spy game, I decided to get all scientific about it and stand up, slowly sliding my water-glass-hand-cup along the wall searching for the optimal audio reception, finding that the studs really do hinder the sound quality, and if I went above the counter level I could get even better audio.
Realizing the compromising position I was in, half squatting with my ear plaster to my hand-cup against the wall and between the exam table and the chair they give you so your clothes aren't just thrown on the floor, I decided that I better just chill out and resume a more natural stance in a section of the room the nurse would not be alarmed to find me.
It wasn't like he'd been telling her anything she's have needed to go all HIPPA on me about. They were discussing what foods constituted a balanced diet. Big whoop. But it was something to do and I tend to do stuff that hidden camera people would EAT UP if I think I'm alone.
About the time I got situated back in my chair where I had been told to sit, stay, was about the same time I heard her door open and the doc saying his goodbyes. Then I could hear him and the nurse discussing his next patient (moi) out in the hallway.
"Oh, they're sisters? Really! Cool."
He then came in, not mentioning my sister or any Family Plan Discount Coupon he might have in his wallet, but just shook my hand and got busy reviewing my file.
He talked about osteopenia and how it shouldn't be confused with osteoporosis since that's not what I have. We talked about how I need to take an aggressive proactive stance against it and start making changes that would decrease my odds of ending up with osteoporosis.
He then asked me how the Estradiol was working for me. I told him my plan to hold off starting until I got the results he'd just given me. He said that was a fair decision. (I didn't know I was getting judged on my decision making).
So now that I was presented with the results it was do or die time. We rehashed all the risk factors checking off those which apply to me..
"Used to smoke, don't have high intake of calcium, sedentary lifestyle, you're thin..."
"Woah! What was that? You think I"m thin?? Can I get that in writing on your watermarked letterhead?"
Then we go on to discuss the percentage of body fat that people have and your optimal body fat in order to hover in a safe place farthest from osteoporosis risk (on the low end) and cardiac complications (on the high end). It needs to be between 25 and 30% on the scale.
Then he tells me there are scales which can accurately calculate body fat and body water, and you can purchase them at wal-mart.com among other places. Intrigued I whipped out some scratch paper and a pen and started taking notes.
Simultaneously he whipped out his iPhone and started surfing to find the one I needed to get, were I so inclined. After writing the brand name (Taylor) and that it needed to be digital and say that it measures body fat and body water, I felt my heart skip a beat...
I had a defective iron I had purchased the day before and needed the receipt to take it back for exchange. I was hoping against hope I wasn't scribbling all over the back of it. I flipped it over and for a second I was relieved.
Then I burst out laughing and confessed to the doctor that I was taking notes about the scale on the back of a Dunkin Donuts receipt. He, too, had a good laugh.
So we decided that I would increase my calcium intake to between 1100 and 1500 mg / day (I have a 1200 mg chewable I got from Walgreens that fits the bill), and I would get that scale, (which I ordered from Wal-mart.com from this ad) , and that I would begin taking the Estradiol as opposed to my inevitable choices I would later be presented with should my bones continue thinning...
Things like Boneva, Fosomax, and other super expensive osteo drugs. I decided I'd give the Estradiol (at $17 for a 1 mo. supply or if I join Walgreens Rx plan I can get it for $4/mo for a 3mo supply) a try. May as well try the cheap route since I'm not made of money last I checked.
That brings us up to date and I am on day 3 of being an estroginated subject.
Here's what I have found.
- 1st day I was a little dizzier than my ususal MS/Fingolimod dizzy.
- I have only gone through 2 nights since starting and already I sleep better with less insomnia, less restless light sleep, more sound sleep and less hot flashes.
- My hips don't hurt as bad. They were stiff and sore all the time but I just figured it was from sitting so much and getting older.
If this helps my menopausal symptoms I had been ignoring (the sleep issues, hot flashes and hip aches) and helps increase my bone density, then I guess I am willing to risk certain minimally possible adverse events.
After all, I'm becoming a pro at this drug taking, weigh the pros and cons stuff. I've had 3 years practice with Fingolimod. :D
Wednesday, September 1, 2010
Another Year Gone By
I know I have really slacked off on the blogging and for those of you eager to hear news about Fingolimod (now Gilenia) I do apologize.
Just know that "no news is good news" is the phrase du jour . My 3 year mark in the clinical trial was on August 20, 2010 - 12 days ago! It still amazes me that I have come so far and feel safe in saying my MS is in remission.
It took me SO LONG to get over the feeling that if I sneezed too hard or crossed my eyes, I'd send myself into a relapse. But before Fingolimod that was exactly the case. It didn't take much at all, even just a little stress, to send me into the land of numbness, burning, tingling and limbs I had to drag.
So I guess I am trying to ignore the fact that the trial is probably winding down, as is sure to happen. They have to wrap this up sometime and with the pending FDA review, I have tried to ignore the fact that I will soon have to figure out how to stay on this medication and maintain my good health.
It's crossed my mind on more than one occasion that maybe all the success I have had isn't TOTALLY attributable to FTY720 alone, but was partly brought about by quitting smoking and caffeine, and trying to make better food choices. This is a comforting idea to ponder because in my little pea brain it suggests that I have a fighting chance of staying in remission even without my drugs.
I think that keeping a positive attitude and having a new outlet for my creative energies has also helped to keep me stable. I just can't get enough of the tie-dyeing that my sister and I have started doing/selling. It's been a joy to have something so satisfyingly artistic to do after years of feeling frustrated that MS had taken my finest motor skills so that I no longer can do really detailed pen and ink drawings.
Tie-dye IS an artform, by gosh! And unlike drawings I may have started and got bored with or frustrated by, none of the tie-dye stuff sits around unfinished. I do them beginning to end with the untying being the most exciting part. It feels like Christmas morning every single time. Love to see the surprising designs of colors and patterns.
It's that exhilaration that keeps us doing it, and the fact that it keeps my mind from stagnating in a MS pondering funk is a great big plus. SELLING something would make me even more ecstatic but I guess that's too much to ask for ROFL.
I have another BIG clinical trial checkup coming soon but oddly I have not heard from my trial nurse in a while. Usually she calls a couple weeks before the appt. to confirm. For the past couple "anniversary" appointments, it's always been a big deal, with every single test performed from PFT to MRI -- all having to be completed within so many days (14 or 30 maybe?) of the anniversary date (8/20).
Counting my pills, I see I have 23 left and I already took today's, so I'm good for a few weeks anyhow.
Until then, I'll keep pretending to be normal and revel in my new found creative outlet. Hopefully I'll have a more MS-y, clinical trial-ish post for you soon.
Just know that "no news is good news" is the phrase du jour . My 3 year mark in the clinical trial was on August 20, 2010 - 12 days ago! It still amazes me that I have come so far and feel safe in saying my MS is in remission.
It took me SO LONG to get over the feeling that if I sneezed too hard or crossed my eyes, I'd send myself into a relapse. But before Fingolimod that was exactly the case. It didn't take much at all, even just a little stress, to send me into the land of numbness, burning, tingling and limbs I had to drag.
So I guess I am trying to ignore the fact that the trial is probably winding down, as is sure to happen. They have to wrap this up sometime and with the pending FDA review, I have tried to ignore the fact that I will soon have to figure out how to stay on this medication and maintain my good health.
It's crossed my mind on more than one occasion that maybe all the success I have had isn't TOTALLY attributable to FTY720 alone, but was partly brought about by quitting smoking and caffeine, and trying to make better food choices. This is a comforting idea to ponder because in my little pea brain it suggests that I have a fighting chance of staying in remission even without my drugs.
I think that keeping a positive attitude and having a new outlet for my creative energies has also helped to keep me stable. I just can't get enough of the tie-dyeing that my sister and I have started doing/selling. It's been a joy to have something so satisfyingly artistic to do after years of feeling frustrated that MS had taken my finest motor skills so that I no longer can do really detailed pen and ink drawings.
Tie-dye IS an artform, by gosh! And unlike drawings I may have started and got bored with or frustrated by, none of the tie-dye stuff sits around unfinished. I do them beginning to end with the untying being the most exciting part. It feels like Christmas morning every single time. Love to see the surprising designs of colors and patterns.
It's that exhilaration that keeps us doing it, and the fact that it keeps my mind from stagnating in a MS pondering funk is a great big plus. SELLING something would make me even more ecstatic but I guess that's too much to ask for ROFL.
I have another BIG clinical trial checkup coming soon but oddly I have not heard from my trial nurse in a while. Usually she calls a couple weeks before the appt. to confirm. For the past couple "anniversary" appointments, it's always been a big deal, with every single test performed from PFT to MRI -- all having to be completed within so many days (14 or 30 maybe?) of the anniversary date (8/20).
Counting my pills, I see I have 23 left and I already took today's, so I'm good for a few weeks anyhow.
Until then, I'll keep pretending to be normal and revel in my new found creative outlet. Hopefully I'll have a more MS-y, clinical trial-ish post for you soon.
Subscribe to:
Posts (Atom)